Reputable factory supply Tetrapropylammonium bromide 1941-30-6 in stock with high standard
- Molecular Formula:C12H28NBr
- Molecular Weight:266.265
- Appearance/Colour:white crystals or crystalline powder
- Melting Point:266-272 °C
- Refractive Index:1.5260 (estimate)
- PSA:0.00000
- Density:1.1949 (rough estimate)
- LogP:0.44720
Tetrapropylammonium bromide(Cas 1941-30-6) Usage
|
Air & Water Reactions
|
Water soluble. Aqueous solutions are weakly acidic.
|
|
Reactivity Profile
|
Tetrapropylammonium bromide is hygroscopic. Tetrapropylammonium bromide is incompatible with strong oxidizing agents. .
|
|
Fire Hazard
|
Flash point data for Tetrapropylammonium bromide are not available; however, Tetrapropylammonium bromide is probably combustible.
|
|
Purification Methods
|
Crystallise it from ethyl acetate/EtOH (9:1), acetone or MeOH. Dry it at 110o under reduced pressure. [Beilstein 4 IV 471.]
|
|
Definition
|
ChEBI: A quarternary ammonium bromide salt in which the cation has four propyl substituents around the central nitrogen.
|
|
General Description
|
White to off-white crystalline solid.
|
InChI:InChI=1/C12H28N.BrH/c1-5-9-13(10-6-2,11-7-3)12-8-4;/h5-12H2,1-4H3;1H/q+1;/p-1
Hebei KuiSheng Trading Co., LTD ,a company specializing in the production and supply of chemicals for various industries. we take pride in our ability to carefully formulate chemicals that meet the highest standards of quality, efficiency, and safety. Through advanced technology and strict quality control measures, we ensure that our products consistently deliver exceptional performance and reliability. Whether you are in need of chemicals for pharmaceutical, agricultural, or industrial applications, we offer a wide range of solutions to meet your specific requirements. Our team is dedicated to providing excellent customer service and we strongly believe in establishing long-lasting business relationships built on trust and mutual success. Allow me to outline some of our key advantages: 1. High quality with competitive prices: We strive to offer chemicals of the highest quality while remaining competitive in the market. 2. All purity >99%: Our products undergo rigorous purification processes to guarantee high purity levels. 3. Manufacturer with factory prices: As a manufacturer, we have the ability to offer high-quality products at competitive factory prices. 4. Fast and safe delivery: We understand the importance of timely and secure deliveries. Therefore, we have established reliable logistics networks to ensure efficient transportation of our products. 5. OEM is welcome: We are open to providing Original Equipment Manufacturer (OEM) services, allowing you to customize products according to your specific needs. 6. Sufficient stock: Our extensive inventory ensures that we can fulfill orders promptly, regardless of their size or complexity. We are confident that with our extensive experience and dedication to excellence, we can be your trusted partner in chemical solutions. We would greatly appreciate the opportunity to work with you and contribute to the success of your business. Should you require any further information or have specific inquiries, please do not hesitate to contact us. We look forward to hearing from you.
1941-30-6 Relevant articles
Solubilization of some Tetramethylammonium Salts and of Ethyltrimethylammonium Bromide by their Homologues in Chloroform
Czapkiewicz, Jan
, p. 2669 - 2674 (1989)
The solubilities of tetramethylammonium ...
An Efficient One-Pot, Four-Component Synthesis of Pyrazolo[3,4-b]pyridines Catalyzed by Tetrapropylammonium Bromide (TPAB) in Water
Ezzati, Mahnaz,Khalafy, Jabbar,Marjani, Ahmad Poursattar,Prager, Rolf H.
, p. 435 - 441 (2018)
A novel and one-pot pseudo-four-componen...
Preferential orientations of structure directing agents in zeolites
Dib, Eddy,Gimenez, Antoine,Mineva, Tzonka,Alonso, Bruno
supporting information, p. 16680 - 16683 (2015/10/05)
The local structure of as-synthesised si...
NMR study of the complex formation between tert-butyl hydroperoxide and tetraalkylammonium bromides
Turovskij, Nikolaj A.,Berestneva, Yulia V.,Raksha, Elena V.,Zubritskij, Mikhail Yu.,Grebenyuk, Serhiy A.
, p. 1443 - 1448 (2014/11/27)
The interaction between tert-butyl hydro...
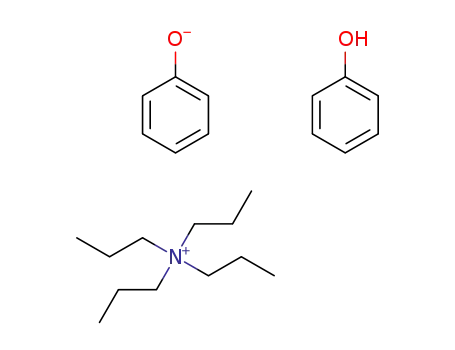
Effects of charge separation, effective concentration, and aggregate formation on the phase transfer catalyzed alkylation of phenol
Denmark, Scott E.,Weintraub, Robert C.,Gould, Nathan D.
supporting information; experimental part, p. 13415 - 13429 (2012/09/25)
The factors that influence the rate of a...
1941-30-6 Process route
-
-
Br(1-)*C12H28N(1+)*C4H10O2
-
- 75-91-2
tert.-butylhydroperoxide
-
- 1941-30-6
tetra-n-propylammonium bromide
Conditions
| Conditions |
Yield |
|
In [D3]acetonitrile; at 24.84 ℃; Equilibrium constant;
|
|
-
- 102-69-2
tri-n-propylamine
-
- 1941-30-6
tetra-n-propylammonium bromide
Conditions
| Conditions |
Yield |
|
In ethanol;
|
|
|
In butanone; at 80 ℃; for 96h;
|
|
|
In acetonitrile; for 24h; Reflux;
|
|
|
In N,N-dimethyl-formamide; at 110 ℃; for 15h; Inert atmosphere;
|
|
1941-30-6 Upstream products
1941-30-6 Downstream products