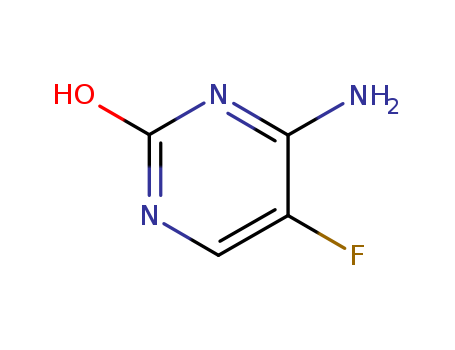
Factory supply Fluorocytosine 2022-85-7 with sufficient stock and high standard
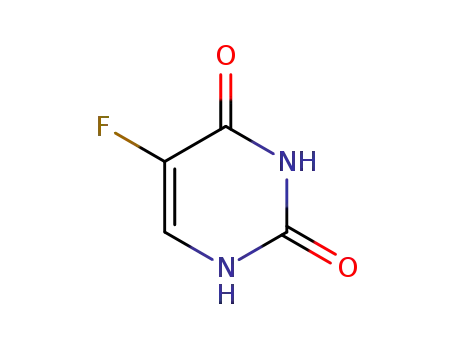
- Molecular Formula:C4H4FN3O
- Molecular Weight:129.094
- Appearance/Colour:white crystalline solid
- Vapor Pressure:0.0492mmHg at 25°C
- Melting Point:298-300 °C (dec.)(lit.)
- Refractive Index:1.613
- Boiling Point:298oC
- PKA:3.26(at 25℃)
- Flash Point:96.4°C
- PSA:71.77000
- Density:1.73 g/cm3
- LogP:0.07240
Hebei KuiSheng Trading Co., LTD ,a company specializing in the production and supply of chemicals for various industries. we take pride in our ability to carefully formulate chemicals that meet the highest standards of quality, efficiency, and safety. Through advanced technology and strict quality control measures, we ensure that our products consistently deliver exceptional performance and reliability. Whether you are in need of chemicals for pharmaceutical, agricultural, or industrial applications, we offer a wide range of solutions to meet your specific requirements. Our team is dedicated to providing excellent customer service and we strongly believe in establishing long-lasting business relationships built on trust and mutual success.
Fluorocytosine(Cas 2022-85-7) Usage
|
Description
|
Fluorocytosine (5-Fluorocytosine or 5-FC) is a synthetic fluorinated pyrimidine, initially developed as an anticancer agent. It was later discovered to be effective against fungal infections, particularly those caused by yeasts like Candida and Cryptococcus species. |
| Mechanism of Action |
Fluorocytosine is converted into 5-fluorouracil (5-FU) by fungal cytosine deaminase, interfering with fungal RNA and DNA synthesis, leading to cell death. |
| Pharmacokinetics |
Absorption: Rapid and nearly complete oral absorption, with a bioavailability of 76-89%.
Distribution: Well distributed in body fluids, including cerebrospinal fluid (CSF) and eye fluids.
Elimination: Primarily excreted via the kidneys, with minimal hepatic metabolism.
Half-life: 3-4 hours in normal renal function; can be extended up to 85 hours in patients with renal insufficiency.
|
| Mechanism of Action |
Once inside susceptible fungal cells, Fluorocytosine is converted to 5-FU, which disrupts:
RNA synthesis by misincorporation, leading to altered protein production.
DNA synthesis by inhibiting thymidylate synthase, a key enzyme in nucleotide production. |
| Side Effects |
Common: Nausea, vomiting, abdominal pain, diarrhea.
Serious: Bone marrow suppression (leukopenia, thrombocytopenia), hepatotoxicity.
Monitoring: Requires close monitoring in patients with renal insufficiency to avoid toxic accumulation.
|
|
Therapeutic Function
|
Antifungal
|
|
Antimicrobial activity
|
The spectrum of activity is restricted to Candida spp., Cryptococcus spp. and some fungi causing chromoblastomycosis.
|
|
Pharmaceutical Applications
|
A synthetic fluorinated pyrimidine available for intravenous infusion or oral administration.
|
|
Drug interactions
|
Potentially hazardous interactions with other drugs Cytarabine: concentration of flucytosine possibly reduced.
|
|
Definition
|
ChEBI: An organofluorine compound that is cytosine that is substituted at position 5 by a fluorine. A prodrug for the antifungal 5-fluorouracil, it is used for the treatment of systemic fungal infections.
|
|
Brand name
|
Ancobon (Valeant).
|
|
General Description
|
Chemical structure: nucleoside
|
InChI:InChI=1/C4H4FN3O/c5-8-2-1-3(6)7-4(8)9/h1-2H,(H2,6,7,9)
Allow me to outline some of our key advantages: 1. High quality with competitive prices: We strive to offer chemicals of the highest quality while remaining competitive in the market. 2. All purity >99%: Our products undergo rigorous purification processes to guarantee high purity levels. 3. Manufacturer with factory prices: As a manufacturer, we have the ability to offer high-quality products at competitive factory prices. 4. Fast and safe delivery: We understand the importance of timely and secure deliveries. Therefore, we have established reliable logistics networks to ensure efficient transportation of our products. 5. OEM is welcome: We are open to providing Original Equipment Manufacturer (OEM) services, allowing you to customize products according to your specific needs. 6. Sufficient stock: Our extensive inventory ensures that we can fulfill orders promptly, regardless of their size or complexity. We are confident that with our extensive experience and dedication to excellence, we can be your trusted partner in chemical solutions. We would greatly appreciate the opportunity to work with you and contribute to the success of your business. Should you require any further information or have specific inquiries, please do not hesitate to contact us. We look forward to hearing from you.
2022-85-7 Relevant articles
Molecular Mechanisms of 5-Fluorocytosine Resistance in Yeasts and Filamentous Fungi
Fatima Zohra Delma 1,Abdullah M. S. Al-Hatmi 2,3,4,Roger J. M. Brüggemann 3,5,Willem J. G. Melchers 1,3,Sybren de Hoog 3,4,Paul E. Verweij 1,3 andJochem B. Buil 1,3,*
, J. Fungi 2021, 7(11), 909;
Effective management and treatment of fungal diseases is hampered by poor diagnosis, limited options for antifungal therapy, and the emergence of antifungal drug resistance. An understanding of molecular mechanisms contributing to resistance is essential to optimize the efficacy of currently available antifungals. In this perspective, one of the oldest antifungals, 5-fluorocytosine (5-FC), has been the focus of recent studies applying advanced genomic and transcriptomic techniques to decipher the order of events at the molecular level that lead to resistance. These studies have highlighted the complexity of resistance and provided new insights that are reviewed in the present paper.
Controlled Synthesis of New 5-Fluorocytosine Cocrystals Based on the pKa Rule
Cecília C. P. da SilvaRebeka de O. PepinoCristiane C. de MeloJuan C. TenorioJavier Ellena*
, Cryst. Growth Des. 2014, 14, 9, 4383–4393
5-Fluorocytosine (5-FC) was investigated for the controlled synthesis of cocrystals by applying the pKa rule. Five cocrystals were designed and developed with adipic, succinic, terephtalic, benzoic, and malic acids, all exhibiting negative ΔpKa values ranging from close to zero up to roughly −1. The synthesized cocrystals were analyzed by single crystal X-ray diffraction, and the observed supramolecular synthons were compared to the reported structures containing 5-FC.
2022-85-7 Upstream products
-
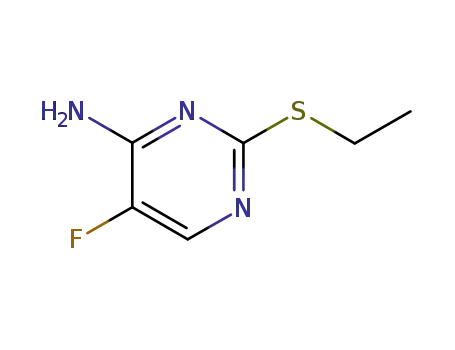
701-87-1
2-ethylsulfanyl-5-fluoro-pyrimidin-4-ylamine
-
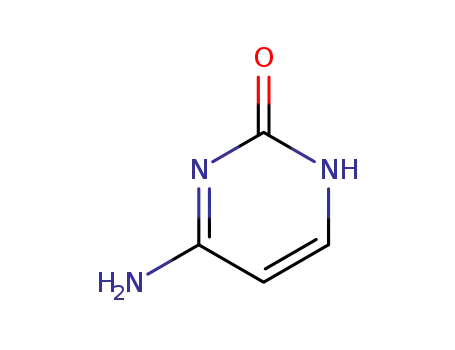
71-30-7
Cytosine
-
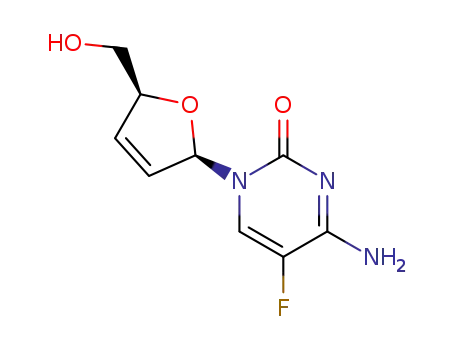
134379-77-4
β-D-2',3'-didehydro-2',3'-dideoxy-5-fluorocytidine
-
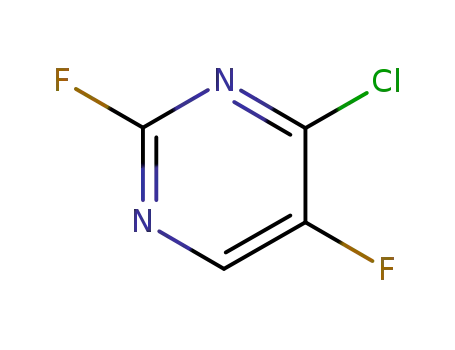
99429-06-8
2,5-difluoro-4-chloro-pyrimidine
2022-85-7 Downstream products
-
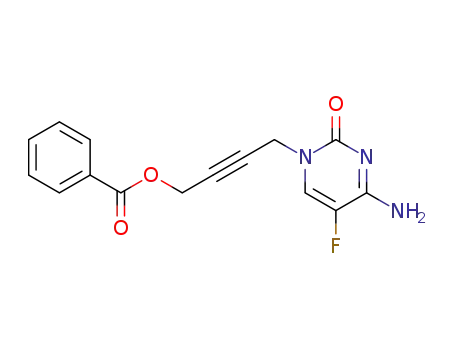
158666-06-9
N1-(4-benzoyl-2-butyn-1-yl)-5-fluorocytosine
-
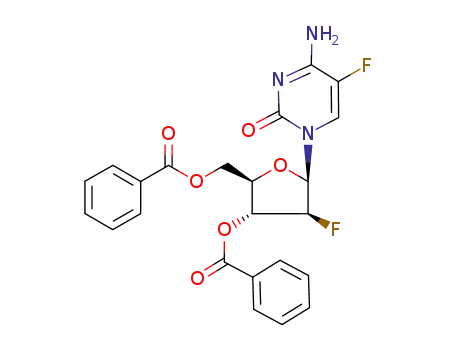
128496-05-9
1-(3,5-O-dibenzoyl-2-fluoro-β-D-arabinofuranosyl)-5-fluorocytosine
-
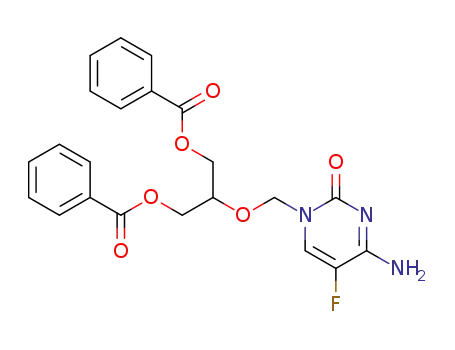
110874-31-2
4-amino-5-fluoro-1-<<2-(benzoyloxy)-1-<(benzoyloxy)methyl>ethoxy>methyl>cytosine
-
51-21-8
5-fluorouracil